Freedom to Operate Search for Medical Device

Launching a new product in regulated industries such as medical technology requires careful evaluation of existing patents. Companies must ensure that their innovations do not infringe active intellectual property rights in the markets where they plan to commercialize their products.
Darsh Analytics supports technology companies by conducting structured Freedom to Operate (FTO) searches that identify potential patent risks before commercialization. Our analysis helps organizations understand the patent landscape, evaluate infringement exposure, and develop strategies to mitigate risks.
In this project, a European MedTech company approached Darsh Analytics while preparing to launch a new medical device in the United States and European markets. The company required a comprehensive assessment of potential patent infringement risks to ensure a smooth product launch and avoid costly legal disputes.

Project Description
A medical technology company based in Europe was preparing to commercialize a new medical device designed for international markets. Before launching the product in the United States and European Union, the company needed to determine whether existing patents could potentially block or restrict their product.
The client required a detailed Freedom to Operate (FTO) analysis focused on the specific product features and the jurisdictions where the product would be launched. The goal was to identify any active patents that could pose infringement risks and provide insights that could guide product design decisions.
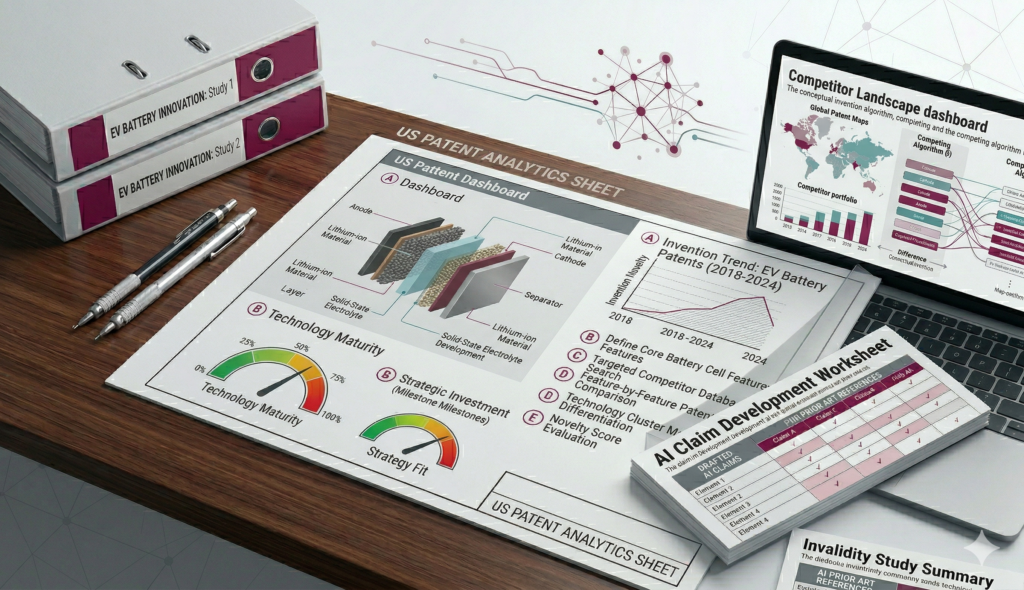
Darsh Analytics conducted a structured FTO search across major patent databases and analyzed the claims of relevant patents in detail. The study identified several active patents related to the technology domain and categorized them based on potential infringement risks.
Our analysis provided the client with clear insights into high‑risk patents and possible design‑around options. This allowed the client to refine certain product features and proceed with commercialization more confidently.
Project Details
- Client Type: Medical Technology Company (Europe)
- Date: 2024
- Service Category: Freedom to Operate (FTO) Search & Analysis
- Technology Domain: Medical Device Technology
- Objective: Assess patent infringement risks before launching in the US and EU markets
Project Participants
- Patent Research Team: Darsh Analytics
- Project Lead: Senior Patent Research Analyst
- Technical Domain Expert: Medical Device Specialist
- Client Team: Product Development Team & Patent Attorney
Challenge
The client planned to introduce a new medical device in the United States and European markets. However, entering these markets without evaluating existing patents could expose the company to infringement litigation, injunctions, or licensing disputes.
The key challenge was to identify active patents related to the product features and determine whether any of them could potentially block commercialization.
Our Approach
Darsh Analytics followed a structured Freedom to Operate methodology:
- Defined the product scope and technical features relevant to the analysis
- Identified target jurisdictions including the United States and European markets
- Conducted searches to identify active patents related to the technology
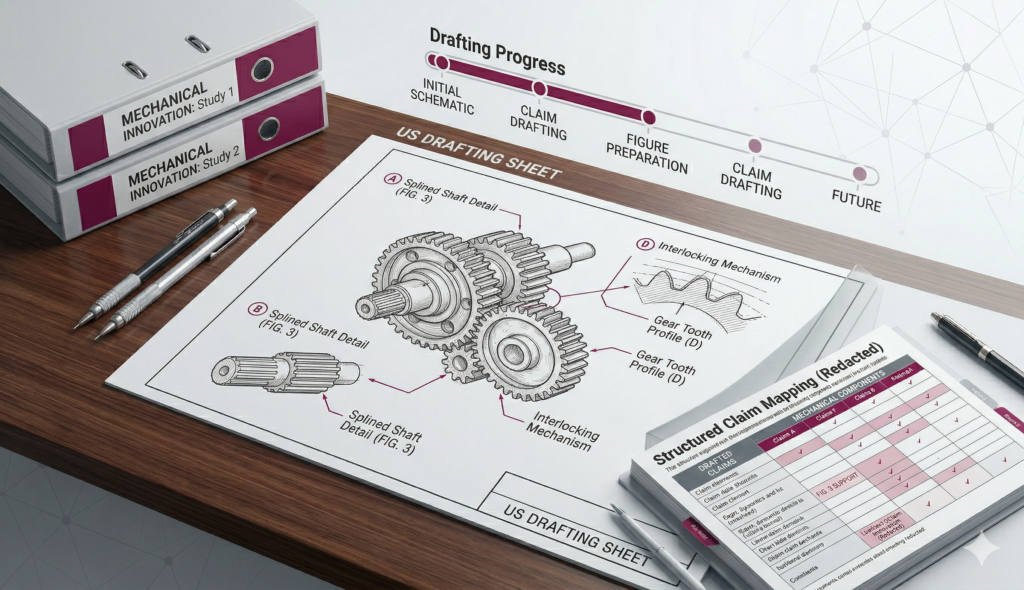
- Performed detailed claim‑level infringement analysis
- Categorized patents based on potential infringement risk
- Suggested possible design‑around considerations where applicable
Outcome
The FTO analysis provided the client with a clear understanding of potential patent risks associated with their product.
As a result:
- High‑risk patents were identified early in the development process
- The client modified specific product design elements
- Potential infringement risks were significantly reduced
- The product successfully entered the target markets with greater legal confidence